It occurs because of the distortion of an electron cloud. In pi bonding, there are two electron clouds present, in which one is above the plane of atomic nuclei, and the other is below it.Unlike sigma bonds, free rotation about pi bonds is not possible.

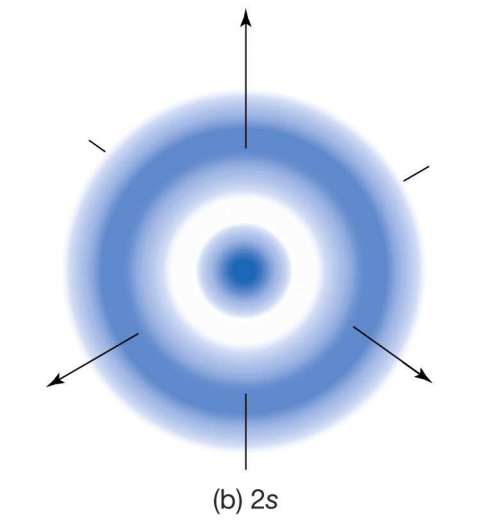
The sideway overlapping of atomic orbitals forms this bond.In sigma bonding, the electron cloud is symmetrical about the internuclear axis, which means it is not localised towards any one atom. Free rotation around the sigma bond is possible.A sigma bond is formed first, then a pi bond because it has lower energy.Sigma bonding orbitals are cylindrically symmetrical orbitals.They are formed by head-on overlapping between the atomic orbitals.Sigma bonds are the strongest covalent bonds.To know the influence of electron clouds on sigma bonds, let us first study sigma bonds. the symmetry of the electronic configuration.Various factors can affect the electron affinity, like: In general, elements with more negative electron affinity are smaller and have the highest ionisation energies. High electron affinity means that the atom can more readily accept electrons, whereas a lower electron affinity indicates that an atom can not accept electrons easily.Įlectron affinity is written with a negative sign, as energy is released in an exothermic process.It is measured in eV/atom, KJ/mol, or Kcal/ mol.It depends on stable electronic configuration it does not regularly increase in a period.It is an energetic term as it describes the released energy of an electron.Electron affinityĮlectron affinity is the energy released when an electron is added to a neutral isolated gaseous atom to form a negatively charged ion. It is formed when the electron clouds of two atoms overlap with each other in a fixed direction. Electron density is used to describe electron electronegativity, and the greater the difference in electronegativity between two atoms, the more the electron density is pulled toward, the more electronegative atom.Electron density is also a term used for describing electron clouds.Because of electron cloud formation, it is impossible to find the simultaneous measurement of the position and momentum of an electron. However, the electron cloud is like a dense cloud of probability that surrounds the nucleus. All these models assumed that an electron is a particle of well-defined mass that revolves around the nucleus like a planetary system. There are always two electrons in every single orbital, which can be different by the property of spin.ĭifferent models first gave a different idea about an atom, like the plum pudding model, Rutherford’s model, Bohr’s model.There are four basic electron orbitals: s, p, d and f.The number and types of orbitals increase with an increase in atomic number.Electron orbitals predict a certain level of probability of the presence of an electron.The electron cloud is a system of electrons surrounding the nucleus of an atom, and each orbital around the nucleus of the atom resembles a cloud-like structure. The higher density of electrons means that the chances of finding an electron are higher, which is why those regions are called electron orbitals. Electrons are most likely to be found in the electron cloud. He developed an equation that can calculate the probability of finding an electron in an area around the nucleus.Īn electron cloud is a group of electrons that circulate the nucleus. Erwin Schrödinger developed the concept of the electron cloud.
